Grams to Moles Calculator
Grams to Moles Calculator
Enter the values below to calculate your chemistry result.
What Is a Mole in Chemistry?
A mole (mol) is the SI base unit for the amount of substance. One mole contains exactly 6.022 × 10²³ elementary entities, whether atoms, molecules, or ions. This fixed number is called Avogadro's constant (NA).
Think of it like a "dozen," but astronomically larger. A dozen always means 12 objects; a mole always means 6.022 × 10²³ objects. What changes between substances is the mass of that mole, and that's what molar mass tells you.
This concept bridges two worlds: the microscopic scale of atoms and the macroscopic world of grams you can measure on a laboratory balance. Without the mole, stoichiometry, solution preparation, and chemical equation balancing would be impossible to do practically.

Key fact: The mole was defined so that one mole of carbon-12 atoms weighs exactly 12 grams. This is why atomic mass units and molar mass are numerically equal.
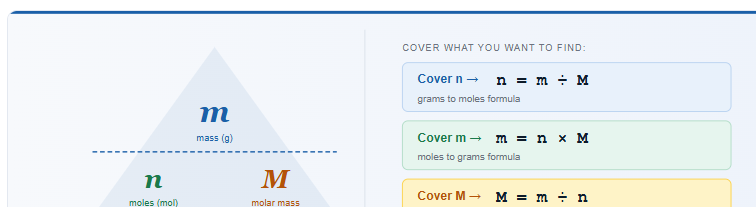
The Grams to Moles Formula (n = m ÷ M)
The relationship between mass, moles, and molar mass is expressed in one equation, the foundation of every stoichiometry calculation in chemistry:
Rearranging gives you the complete set of three formulas:
| Find | Formula | When to Use |
|---|---|---|
| Moles (n) | n = m ÷ M | You have mass, want moles |
| Mass in grams (m) | m = n × M | You have moles, want mass |
| Molar mass (M) | M = m ÷ n | You have both mass and moles |
How to Convert Grams to Moles — Step by Step
Worked Examples: Grams to Moles (and Back)
Follow these complete solutions to understand the method, not just the answer.
How many moles are in 36 g of water?
→ Molar mass of H₂O = 2(1.008) + 15.999 = 18.015 g/mol
→ n = 36 ÷ 18.015
What is the mass of 0.5 mol of NaCl?
→ Molar mass of NaCl = 22.99 + 35.45 = 58.44 g/mol
→ m = 0.5 × 58.44
How many moles are in 1 kg of glucose?
→ Convert: 1 kg = 1,000 g
→ Molar mass = 6(12.01) + 12(1.008) + 6(16.00) = 180.16 g/mol
→ n = 1000 ÷ 180.16
A 180.16 g sample contains 2.0 mol of a compound. What is its molar mass?
→ M = m ÷ n = 180.16 ÷ 2.0
Particle count from moles: Multiply moles by Avogadro's number: N = n × 6.022 × 10²³. For example, 2 mol of water = 1.204 × 10²⁴ molecules.
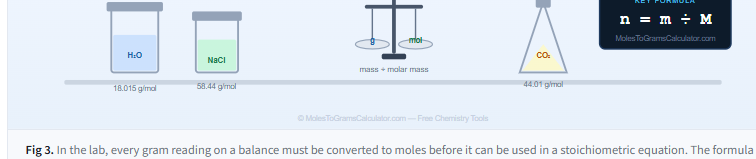
Molar Mass Reference Table for Common Substances
Quickly look up the molar mass of compounds commonly used in homework, lab prep, and stoichiometry problems.
| Substance | Formula | Molar Mass (g/mol) | Bond Type |
|---|---|---|---|
| Water | H₂O | 18.015 | Covalent |
| Sodium chloride (table salt) | NaCl | 58.44 | Ionic |
| Carbon dioxide | CO₂ | 44.01 | Covalent |
| Glucose (blood sugar) | C₆H₁₂O₆ | 180.16 | Covalent |
| Ammonia | NH₃ | 17.03 | Covalent |
| Sulfuric acid | H₂SO₄ | 98.08 | Covalent |
| Calcium carbonate | CaCO₃ | 100.09 | Ionic |
| Ethanol | C₂H₅OH | 46.07 | Covalent |
| Sodium hydroxide | NaOH | 40.00 | Ionic |
| Hydrochloric acid | HCl | 36.46 | Covalent |
| Oxygen gas | O₂ | 32.00 | Elemental |
| Carbon | C | 12.01 | Elemental |
Common Mistakes Students Make (And How to Avoid Them)
Why the Grams-to-Moles Conversion Matters in Real Chemistry
Stoichiometry and Balanced Equations
Balanced chemical equations express reactions in mole ratios, not gram ratios. Before calculating yield or weighing out a reactant, every mass must be converted to moles.
Preparing Solutions (Molarity)
When a protocol calls for a 0.1 M solution, that means 0.1 mol of solute per litre. To weigh the right amount, you convert those 0.1 moles into grams using m = n × M.
Pharmaceutical and Industrial Applications
Every active pharmaceutical ingredient is measured with molecular precision. Manufacturers convert a target mole count into grams to determine the mass per batch. The same logic governs fertiliser production, polymer chemistry, and environmental analysis.
Lab note: An error in this conversion doesn't just hurt your grade. In a real lab it can ruin an entire experiment. Always verify by working backwards.
Frequently Asked Questions
Click any question to expand the answer.
How do you convert grams to moles?
Divide the mass in grams by the molar mass in g/mol: n = m ÷ M. For example, 36 g of water ÷ 18.015 g/mol = 2.00 mol.
What is molar mass and how do I calculate it?
Molar mass is the mass of one mole of a substance. Calculate it by adding the atomic masses of every atom in the formula.
What is Avogadro's number and why does it matter?
Avogadro's number is 6.022 × 10²³ particles per mole. It connects measurable grams to particle counts.
Can moles be a decimal or fraction?
Yes. Moles often appear as decimals, such as 0.25 mol or 2.50 mol.
What is the difference between molar mass and molecular weight?
They are numerically similar for many chemistry problems, but molar mass uses g/mol while molecular weight is usually dimensionless.
How do I handle hydrated compounds like CuSO₄·5H₂O?
Include the water molecules in the molar mass. Add the mass of CuSO₄ plus five water molecules.
My mass is in milligrams. How do I use this calculator?
Convert milligrams to grams first by dividing by 1,000.
How many moles are in 100 g of water?
100 g ÷ 18.015 g/mol = 5.55 mol of water.
Key Takeaways
- 1 mole = 6.022 × 10²³ particles.
- Grams to moles: n = m ÷ M.
- Moles to grams: m = n × M.
- Molar mass is the sum of all atomic masses in the formula.
- Always confirm mass is in grams before calculating.
- This conversion is the foundation of every stoichiometry problem.